Cardiovascular
FDA Breakthrough Device Designation
Cardiovascular
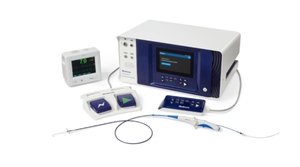
Pi-Cardia’s ShortCut Catheter Nabs FDA Breakthrough Device DesignationPi-Cardia’s ShortCut Catheter Nabs FDA Breakthrough Device Designation
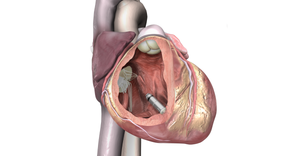
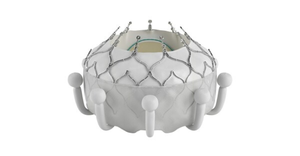
The catheter is the first dedicated leaflet modification device to enable TAVR in patients at risk of coronary obstruction.
Sign up for the QMED & MD+DI Daily newsletter.

.png?width=700&auto=webp&quality=80&disable=upscale)
.png?width=700&auto=webp&quality=80&disable=upscale)
.png?width=700&auto=webp&quality=80&disable=upscale)





.png?width=300&auto=webp&quality=80&disable=upscale)


.png?width=300&auto=webp&quality=80&disable=upscale)